

8:30 am EDT - 2:30 pm EDT
Past Event
8:30 am - 2:30 pm EDT
1775 Massachusetts Avenue NW
Washington, DC
20036
On July 19, 2013, the Engelberg Center for Health Care Reform at Brookings convened an expert workshop to explore strategies to reduce the purchase and use of counterfeit and unapproved drugs in clinical settings. The U.S. Food and Drug Administration (FDA) has undertaken extensive efforts to ensure the integrity of the pharmaceutical supply chain; however, the purchase and use of counterfeit and unapproved drugs in clinical settings (e.g., hospitals, physician’s offices) has become an increasing problem that is neither well understood, nor within the traditional scope of the FDA’s regulatory authority. These illegal and unregulated products cause a serious public health concern as they may contain dangerous substances, incorrect dosages, or no active ingredient. It is clear that combating the emergence of counterfeit and unapproved medications within clinical settings will require broad stakeholder input from FDA, providers, administrators, payers, patients, and others. To this end, the Engelberg Center, in collaboration with FDA’s Office of Drug Security, Integrity, and Recalls, brought together a diverse set of stakeholders in a neutral forum to understand existing approaches to controlling counterfeit and unapproved drugs, identify strategies to improve communication about and awareness of this topic among stakeholders, and develop creative, feasible solutions to deter the use of counterfeit and unapproved products.
9:15 am
10:45 am
1:00 pm
9:30 am

Wendell Primus
March 25, 2026
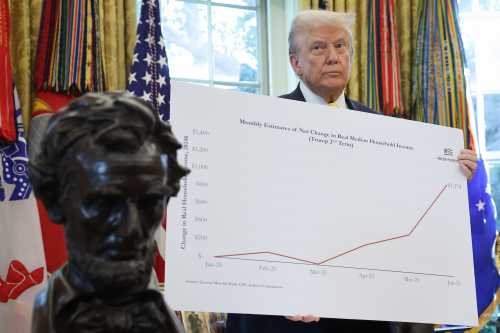
William A. Galston
January 15, 2026

Wendell Primus, Ben Graham, Chloe Zilkha, Sonia Jain
January 6, 2026