

9:00 am EST - 3:00 pm EST
Past Event
9:00 am - 3:00 pm EST
1775 Massachusetts Ave., N.W.
Washington, DC
20036
On November 4, the Engelberg Center for Health Care Reform convened an expert workshop “Exploring Implications of the Nonprescription Drug Safe Use Regulatory Expansion (NSURE) Initiative on Reimbursement and Access.”
In an effort to address the undertreatment of common diseases and conditions, the U.S. Food and Drug Administration is exploring how a regulatory expansion of the nonprescription drug class might increase access to medications. This initiative, known as the Nonprescription Safe Use Regulatory Expansion (NSURE), is exploring how health care professionals and innovative technologies can serve as a condition to the safe use of drugs in a nonprescription setting.
Understanding the impact of NSURE on reimbursement and patient access to medication will be critical to the initiative’s success in addressing medical undertreatment. At this expert workshop, a wide range of experts and stakeholders explored previous effects from prescription-to-nonprescription medication switches, identified potential incentives for coverage of nonprescription drugs with Conditions of Safe Use, and investigated potential mechanisms for reimbursement.
This expert workshop was the third in a series of meetings that explored the NSURE initiative. To explore previous discussions regarding the potential role of health care professionals as a Condition of Safe Use within the NSURE initiative, please visit our expert workshop, “Nonprescription Medications with Conditions of Safe Use as a Novel Solution for Undertreated Diseases or Conditions”. To explore previous discussion regarding the role of technology in supporting the safe and effective use of nonprescription products, please visit our expert workshop, “Innovative Technologies and Nonprescription Medications: Addressing Undertreated Diseases and Conditions through Technology Enabled Self-Care”.
11:00 am
1:00 pm
9:15 am
9:45 am

Wendell Primus
March 25, 2026
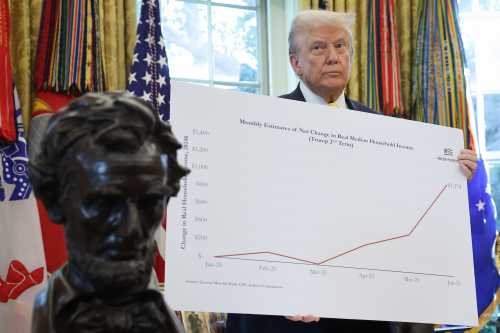
William A. Galston
January 15, 2026

Wendell Primus, Ben Graham, Chloe Zilkha, Sonia Jain
January 6, 2026